P-2936
Lebrikizumab provides high levels of effectiveness in patients with moderate-to-severe atopic dermatitis in routine practice: interim analysis of the non-interventional study AD-LIFE
Felix Lauffer1, Ralf Gutzmer2, Franca Wiemers3, Tobias Hagemann4, Laura Susok5, Afra Kempf6, Sebastian Diemert6, Sven Silberborth6, Diamant Thaçi7
1 Department of Dermatology and Allergy, LMU Hospital, Munich, Germany; 2 Johannes Wesling University Medical Center, Minden, Germany; 3 Dermatology Practice of Dr. med. Wiemers, Leipzig, Germany; 4 Private Dermatological Practice of Dr. med. Tobias Hagemann, Bad Sassendorf, Germany; 5 Department of Dermatology, Dortmund Hospital, University Witten/Herdecke, Germany; 6 Almirall Hermal GmbH, Hamburg, Germany; 7 Institute and Comprehensive Center for Inflammatory Medicine at the University of Lübeck, Lübeck, Germany
Almirall, S.A. has licensed the rights to develop and commercialize lebrikizumab for the treatment of dermatology indications, including atopic dermatitis, in Europe. Lilly has exclusive rights for the development and commercialization of lebrikizumab in the United States and the rest of the world outside of Europe.
EADV Congress; Paris, France; September 17 – 20, 2025
BACKGROUND & OBJECTIVE
- Lebrikizumab (LEB) is a monoclonal antibody that selectively targets IL-13 with high affinity and slow dissociation rate, preventing the IL-13Rα1/IL-4Rα complex formation, and blocking downstream signaling, while avoiding broader immunosuppression.1
- Efficacy and safety of lebrikizumab (LEB) for the treatment of moderate-to-severe atopic dermatitis (AD) was demonstrated throughout its extensive clinical development program.2-4
- However, real-world data on LEB in AD remains scarce as of today.
- The aim of this analysis is to report interim effectiveness and safety data from AD-Life (an open, prospective, multi-center, observational study conducted in Germany to assess AD control at Week [W] 52).
- This is the first study reporting real-world evidence on LEB for moderate-to-severe AD.
CONCLUSION
-
This interim results show that LEB provides high effectiveness with relevant improvements of signs and symptoms in patients with AD, and a favorable safety profile.
- At W16 more than 60% of patients achieved EASI75 and a 4-point-improvement of peak-pruritus, respectively.
- Clinically relevant improvements were seen within the first 4W of treatment.
- Concomitant use of TCS as well as their potency decreased from baseline to W16. - These data suggest that efficacy seen in randomized clinical trials translates well into the real-world setting even with a significant share of patients being pre-treated with advanced systemics and patients suffering from comorbid conditions.
KEY FINDINGS
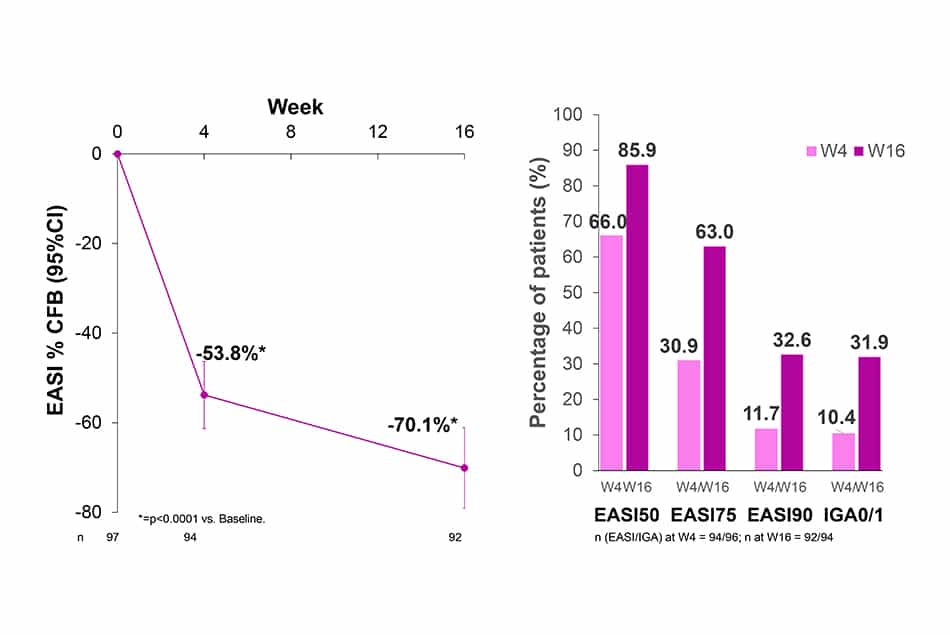
Clinical signs of AD (EASI and IGA)
- After 4 and 16 weeks of treatment with LEB Q2W, EASI decreased by 53.8% and 70.1%, respectively (p<0.0001).
- 85.9%, 63.0%, 32.6% and 31.9% of patients treated with LEB Q2W achieved EASI50, EASI75, EASI90 and IGA0/1, respectively at W16.
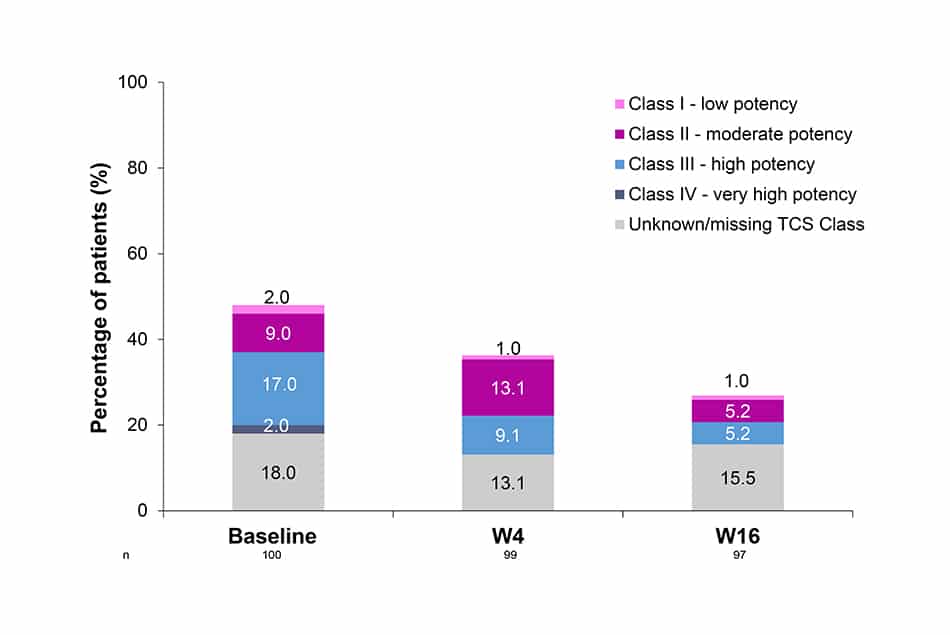
Concomitant use of TCS
- The use of TCS through 16W was reduced: 48% of patients were using them at baseline vs 26% by W16, alongside the lower potency of TCS used in this same period.
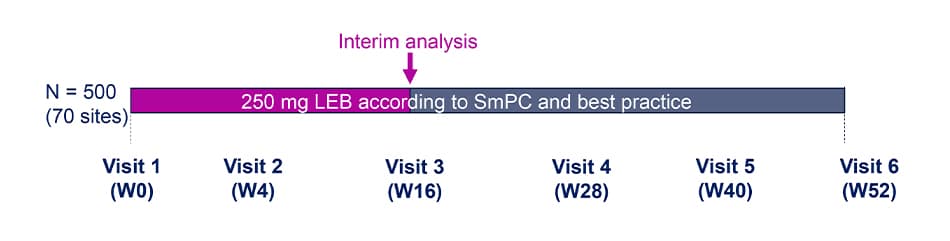
Study Design
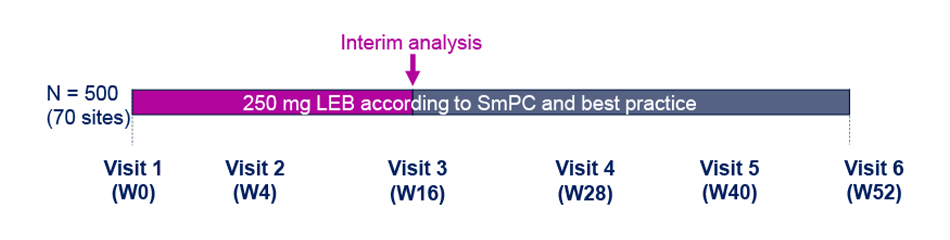
- This is a prospective, multi-center, observational cohort study to evaluate AD control at W52.
- The clinical decision for treatment with LEB is taken by the investigators independently of the participation in the study and prior to the informed consent process.
Key eligibility criteria
Inclusion criteria
- Adult patients ≥ 18 years of age
- Moderate-to-severe AD requiring systemic therapy
- Treatment with LEB according to physician’s decision and in line with the SmPC
- Consented and ready to read and understand study documents.
Exclusion criteria
- Contraindications according to the current SmPC
- Current participation in any interventional clinical trial.
Abbreviations: AD=Atopic Dermatitis; ADR=Adverse Drug Reaction; BMI=Body Mass Index; BSA=Body Surface Area; CFB=Change From Baseline; DLQI=Dermatology Life Quality Index; EASI=Eczema Area and Severity Index; IGA=Investigator Global Assessment; LEB=Lebrikizumab; NRS=Numerical Rate Scale; POEM=Patient-Oriented Eczema Measure; Q2W=Every 2 Weeks; SD=Standard Deviation; SmPC=Summary of Product Characteristics; TCS=Topical Corticosteroids; W=Week.
Population and Analysis
Analysis population
- This interim analysis included patients completing 16W of treatment. Patients had to have either a documented visit at W16 or a confirmed early termination within the first 16W of the study.
Selected Effectiveness Outcomes
- Percent change from baseline (CFB) of EASI up to W16
- Percentage of patients achieving EASI50, EASI75, EASI90 and IGA0/1 over time (W16)
- Percent CFB of peak pruritus numerical rate scale (peak-pruritus NRS)
- Percentage of patients with peak-pruritus NRS ≥4-point improvement from baseline (W4 and W16).
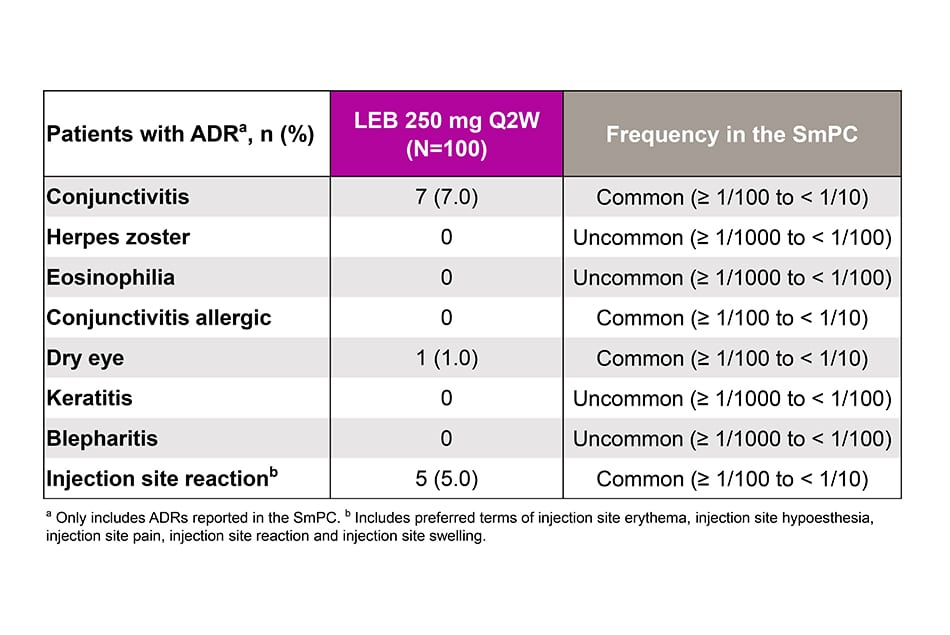
Safety Outcomes
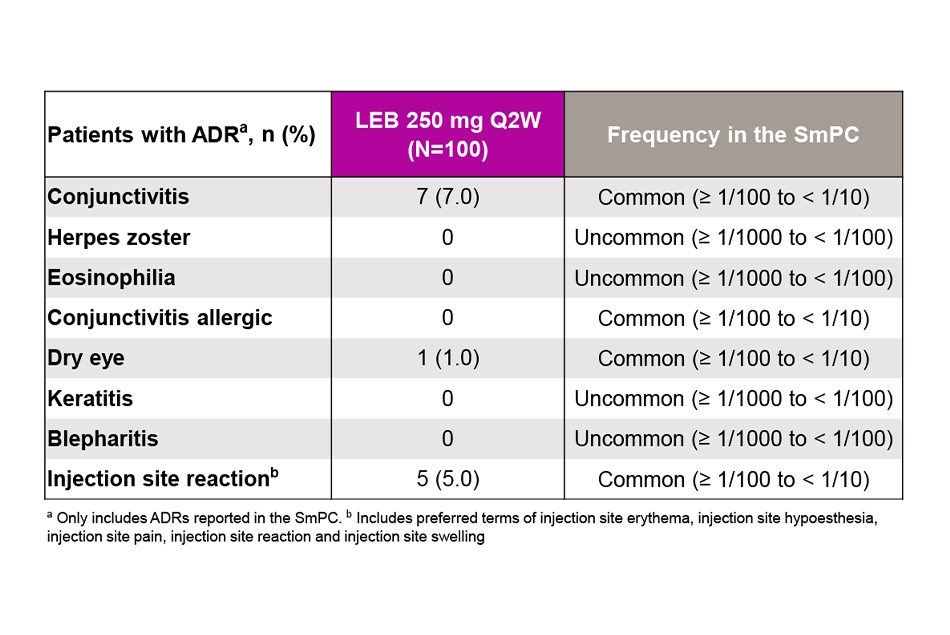
- Adverse drug reactions (ADRs).
Statistical analysis
- Data are reported as observed
- One-sample T-Test was performed for CFB analyses.
Acknowledgments: The authors would like to thank TFS HealthScience for their writing and editorial contributions. This study was funded by Almirall S.A.
References
References
- Okragly AJ, et al. Dermatol Ther (Heidelb). 2023;13:1535-47.
- Silverberg JI, et al. N Engl J Med. 2023;388:1080-91.
- Thaçi D, et al. EADV Congress; 2024; Amsterdam, Netherlands. Poster D1T01.2
- Stein Gold L, et al. Am J Clin Dermatol. 2023;24:595-607.
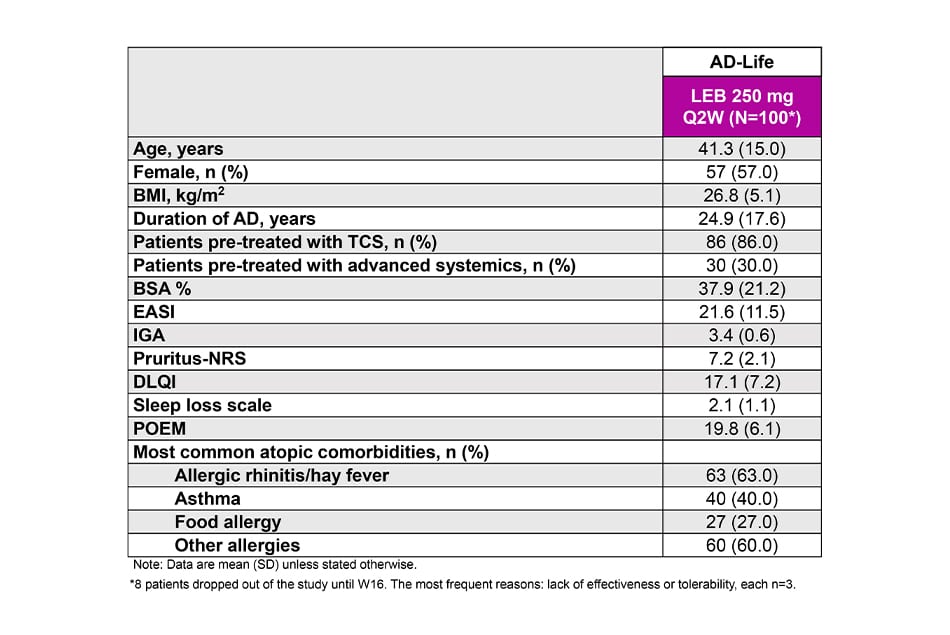
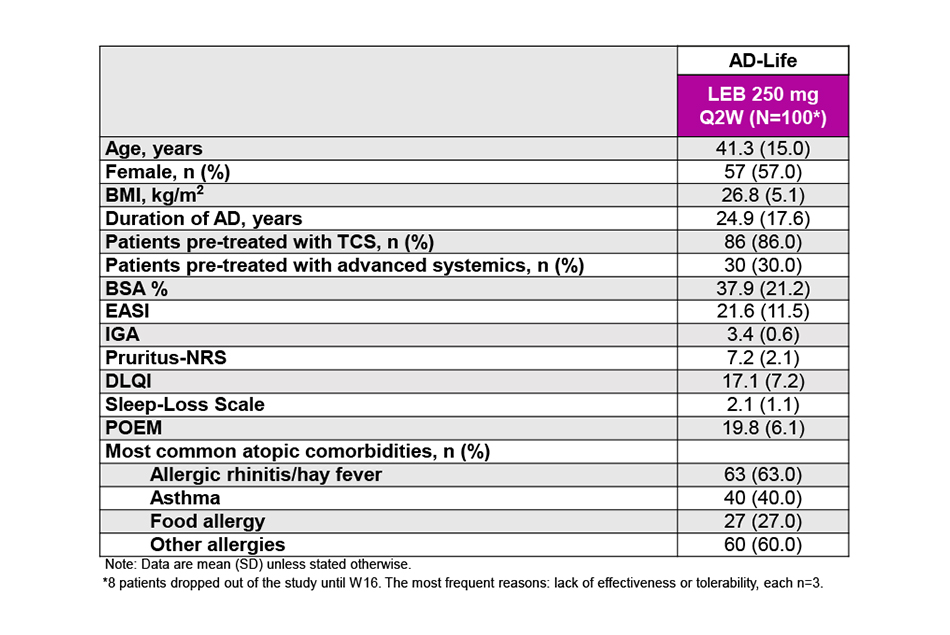
Baseline demographics and disease severity
Disclosures: FL advisor and/or received speaker’s honoraria of the following companies: Abbvie, Almirall, Amgen, Biogen, Boehringer Inglheim, Bristol-Myers-Squibb, Janssen, LEO Pharma, Lilly, Novartis, Roche, Sanofi, Regeneron, UCB, Union Therapeutics, Hexal, Biogen. RG: Research support (to institution): SUN Pharma, Amgen, Sanofi/Regeneron, Merck-Serono, Almirall-Hermal, Kyowa-Kirin, Recordati; Honoraria for lectures/advice: Bristol-MyersSquibb, Novartis, MSD, Almirall-Hermal, Merck-Serono, SUN Pharma, Pierre-Fabre, Sanofi/Regeneron, Immunocore, and Delcath; Meeting support: Pierre-Fabre, SUN Pharma. FW did not have conflicts of interest to disclose. LS has received speakers and/or advisory board honoraria from MSD Sharp&Dohme, Almirall Hermal, Boehringer Ingelheim, Sanofi, UCB, Pfizer, Abbvie, LEO-Pharma. TH has received honoraria as an advisor, speaker and/or investigator from AbbVie, Almirall and Janssen. AK, SD and SS are employees of Almirall Hermal GmbH. DT has received honoraria as an advisor, speaker and/or investigator from AbbVie, Amgen, Almirall, Bristol-Myers-Squibb, Boehringer Ingelheim, Celltrion, Fresenius, Eli Lilly and Company, Galderma, Genzyme, Johnsson& Johnsson, LEO Pharma, New Bridge, Novartis, Pfizer, Regeneron, La Roche-Posay Sanofi, Samsung, Sun Pharma, UCB and Vichy.p;
P-3457
Patient reported outcomes in lebrikizumab treated patients with moderate-to-severe atopic dermatitis in a real-world-setting: interim analysis of the non-interventional study AD-LIFE
Felix Lauffer1, Ralf Gutzmer2, Franca Wiemers3, Tobias Hagemann4, Laura Susok5, Afra Kempf6, Sebastian Diemert6, Sven Silberborth6, Diamant Thaçi7
1 Department of Dermatology and Allergy, LMU Hospital, Munich, Germany; 2 Johannes Wesling University Medical Center, Minden, Germany; 3 Dermatology Practice of Dr. med. Wiemers, Leipzig, Germany; 4 Private Dermatological Practice of Dr. med. Tobias Hagemann, Bad Sassendorf, Germany; 5 Department of Dermatology, Dortmund Hospital, University Witten/Herdecke, Germany; 6 Almirall Hermal GmbH, Hamburg, Germany; 7 Institute and Comprehensive Center for Inflammatory Medicine at the University of Lübeck, Lübeck, Germany
Almirall, S.A. has licensed the rights to develop and commercialize lebrikizumab for the treatment of dermatology indications, including atopic dermatitis, in Europe. Lilly has exclusive rights for the development and commercialization of lebrikizumab in the United States and the rest of the world outside of Europe.
EADV Congress; Paris, France; September 17 – 20, 2025
BACKGROUND & OBJECTIVE
- Lebrikizumab (LEB) is a monoclonal antibody that selectively targets IL-13 with high affinity and slow dissociation rate, preventing the IL-13Rα1/IL-4Rα complex formation, and blocking downstream signaling, while avoiding broader immunosuppression.1
- Efficacy and safety of LEB for the treatment of moderate-to-severe atopic dermatitis (AD) was demonstrated throughout its extensive clinical development program.2-4
- Moreover, LEB treatment provided clinically meaningful improvements in outcomes based on health-related quality of life (HRQoL) and mental health for patients with moderate-to-severe AD in the randomized trials.5, 6
- Real-world data on LEB in AD remains scarce as of today.
- Here, we report interim patient-reported outcomes (PROs) and safety data from AD-Life (an open, prospective, multi-center, observational study conducted in Germany to assess AD control at Week [W)] 52).
- This is the first study reporting real-world evidence on LEB for moderate-to-severe AD.
CONCLUSION
-
LEB showed relevant improvements in PROs in AD within the first 16W of treatment, with a favorable safety profile.
- Approximately 6 out of ten patients reported no or only small impact on HRQoL (DLQI ≤5) at W16 and more than 30% reported no impact on HRQoL at all
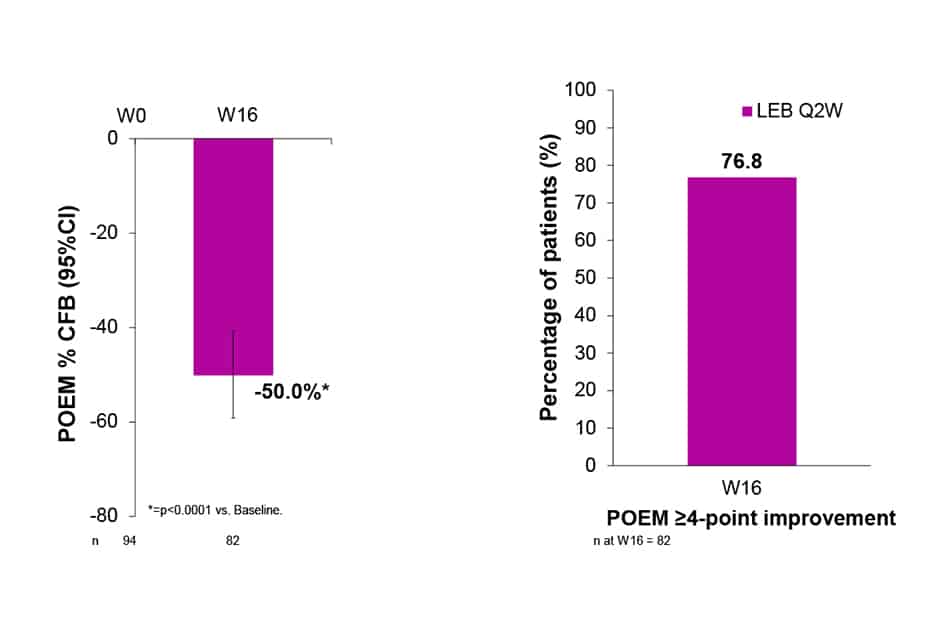
- More than 3 out of 4 patients reported a clinically meaningful improvement of ≥ 4-points in POEM score at W16
- Around 7 out of 10 patients treated with LEB experienced at least 1-point improvement in sleep loss at W16, with relevant results observed at W4 - These findings provide reassurance on the clinical benefits of LEB to patients in a real-world setting in a population with AD suffering from several comorbidities and with patients priorly treated with other advanced systemics.
KEY FINDINGS
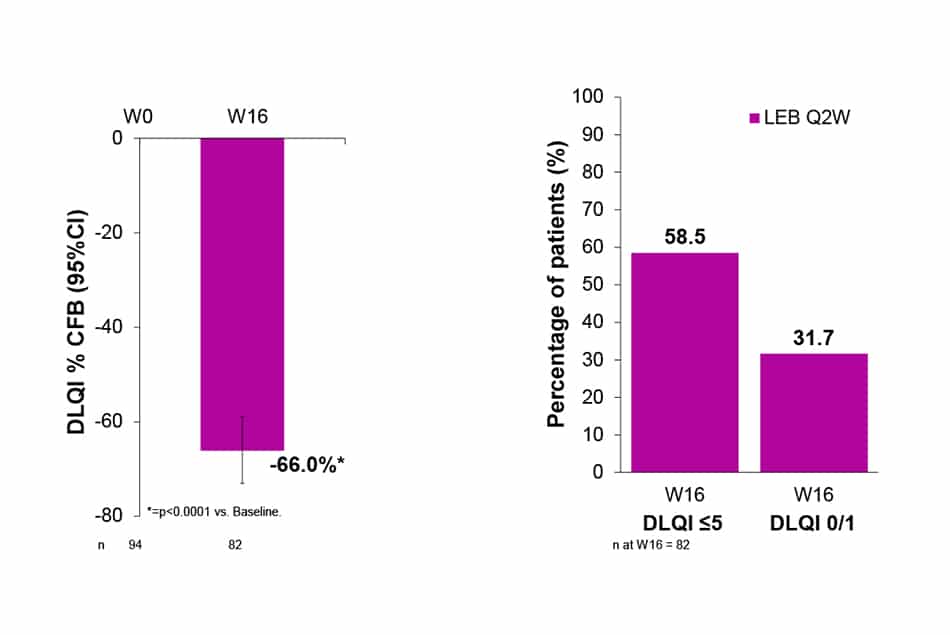
Dermatology Life Quality Index (DLQI)
- DLQI was decreased by 66.0% (p<0.0001) at W16 of treatment with LEB Q2W.
- At W16, 58.5% of patients had DLQI ≤5 and 31.7% achieved DLQI 0/1.
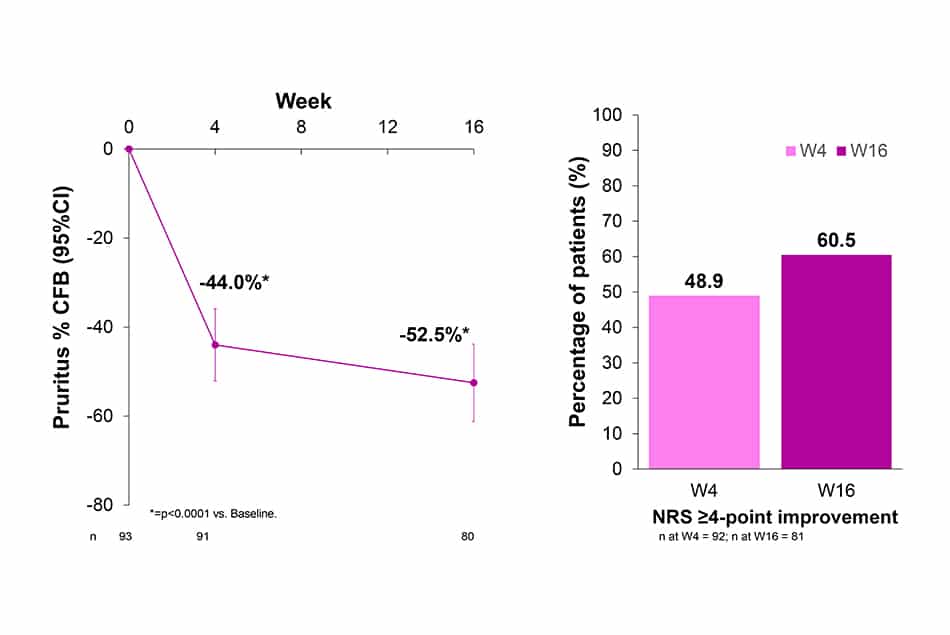
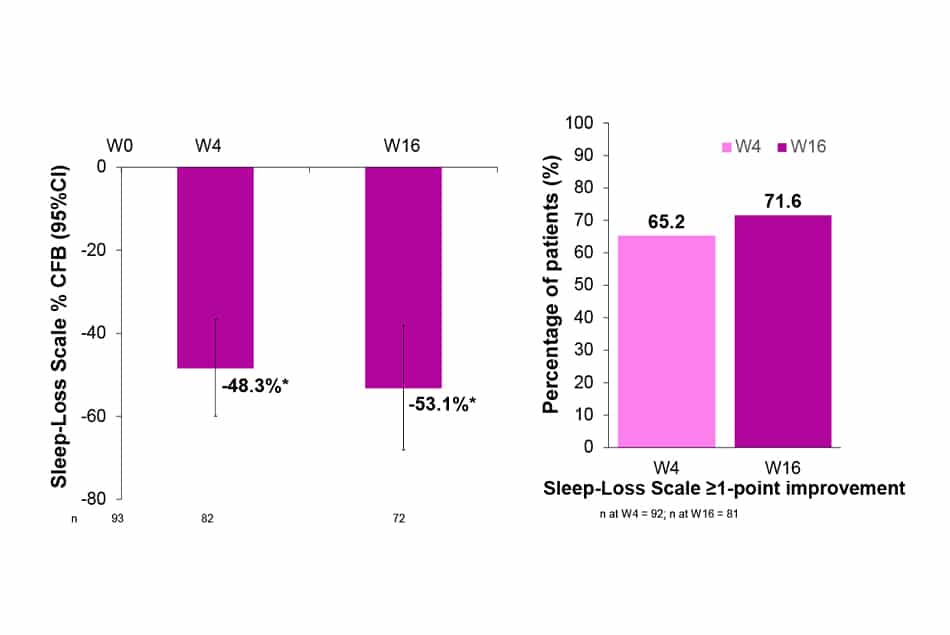
Sleep-Loss Scale / Peak Pruritus-NRS
- After 4 weeks of treatment with LEB Q2W, baseline sleep-loss was improved by 48.3%. This improvement was further increased up to 53.1% (p<0.0001) at W16.
- 65.2% and 71.6% of patients treated with LEB Q2W presented sleep-loss ≥1-point improvement from baseline at W4 and W16, respectively.
- Peak Pruritus-NRS with ≥4-point improvement from baseline was achieved by 48.9% and 60.5% of patients at W4 and W16, respectively (data not shown).
Study Design
- This is a prospective, multi-center, observational cohort study to evaluate AD control at W52.
- The clinical decision for treatment with LEB is taken by the investigators independently of the participation in the study and prior to the informed consent process.
Key eligibility criteria
Inclusion criteria
- Adult patients ≥ 18 years of age
- Moderate-to-severe AD requiring systemic therapy
- Treatment with LEB according to physician’s decision and in line with the SmPC
- Consented and ready to read and understand study documents.
Exclusion criteria
- Contraindications according to the current SmPC
- Current participation in any interventional clinical trial.
Abbreviations: AD=Atopic Dermatitis; ADR=Adverse Drug Reaction; BMI=Body Mass Index; BSA=Body Surface Area; CFB=Change From Baseline; DLQI=Dermatology Life Quality Index; EASI=Eczema Area and Severity Index; IGA=Investigator Global Assessment; LEB=Lebrikizumab; NRS=Numerical Rate Scale; POEM=Patient-Oriented Eczema Measure; Q2W=Every 2 Weeks; SD=Standard Deviation; SmPC=Summary of Product Characteristics; TCS=Topical Corticosteroids; W=Week.
Population and Analysis
Analysis population
- This interim analysis included patients completing 16W of treatment. Patients had to have either a documented visit at W16 or a confirmed early termination within the first 16W of the study.
Patient Reported Outcomes
- Percent change from baseline (CFB) of Dermatology Life Quality Index (DLQI)
- Percentage of patients with DLQI ≤5%
- Percentage of patients with DLQI 0/1
- CFB of Patient-Oriented Eczema Measure (POEM)
- Percentage of patients with POEM ≥4-point improvement from baselinePercentage of patients with POEM ≥4-point improvement from baseline
- Percent CFB of Sleep-Loss Scale (sleep-loss)
- Percentage of patients with sleep-loss ≥1-point improvement from baseline (W16)
- Percentage of patients with peak-pruritus NRS ≥4-point improvement from baseline (W4 and W16).
Safety Outcomes
- Adverse drug reactions (ADRs).
Statistical analysis
- Data are reported as observed
- One-sample T-Test was performed for CFB analyses.
Acknowledgments: The authors would like to thank TFS HealthScience for their writing and editorial contributions. This study was funded by Almirall S.A.
Baseline demographics and disease severity
References
Disclosures:: FL advisor and/or received speaker’s honoraria of the following companies: Abbvie, Almirall, Amgen, Biogen, Boehringer Inglheim, Bristol-Myers-Squibb, Janssen, LEO Pharma, Lilly, Novartis, Roche, Sanofi, Regeneron, UCB, Union Therapeutics, Hexal, Biogen. RG: Research support (to institution): SUN Pharma, Amgen, Sanofi/Regeneron, Merck-Serono, Almirall-Hermal, Kyowa-Kirin, Recordati; Honoraria for lectures/advice: Bristol-MyersSquibb, Novartis, MSD, Almirall-Hermal, Merck-Serono, SUN Pharma, Pierre-Fabre, Sanofi/Regeneron, Immunocore, and Delcath; Meeting support: Pierre-Fabre, SUN Pharma. FW did not have conflicts of interest to disclose. LS has received speakers and/or advisory board honoraria from MSD Sharp&Dohme, Almirall Hermal, Boehringer Ingelheim, Sanofi, UCB, Pfizer, Abbvie, LEO-Pharma. TH has received honoraria as an advisor, speaker and/or investigator from AbbVie, Almirall and Janssen. AK, SD and SS are employees of Almirall Hermal GmbH. DT has received honoraria as an advisor, speaker and/or investigator from AbbVie, Amgen, Almirall, Bristol-Myers-Squibb, Boehringer Ingelheim, Celltrion, Fresenius, Eli Lilly and Company, Galderma, Genzyme, Johnsson& Johnsson, LEO Pharma, New Bridge, Novartis, Pfizer, Regeneron, La Roche-Posay Sanofi, Samsung, Sun Pharma, UCB and Vichy.
- Okragly AJ, et al. Dermatol Ther (Heidelb). 2023;13:1535-47.
- Silverberg JI, et al. N Engl J Med. 2023;388:1080-91.
- Thaçi D, et al. EADV Congress; 2024; Amsterdam, Netherlands. Poster D1T01.2
- Stein Gold L, et al. Am J Clin Dermatol. 2023;24:595-607.
- Lio PA, et al. Dermatol Ther (Heidelb). 2024;14;1929-43.
- Yosipovitch G, et al. Dermatol Ther (Heidelb). 2024;14:2171-80.
Disclosures:: FL advisor and/or received speaker’s honoraria of the following companies: Abbvie, Almirall, Amgen, Biogen, Boehringer Inglheim, Bristol-Myers-Squibb, Janssen, LEO Pharma, Lilly, Novartis, Roche, Sanofi, Regeneron, UCB, Union Therapeutics, Hexal, Biogen. RG: Research support (to institution): SUN Pharma, Amgen, Sanofi/Regeneron, Merck-Serono, Almirall-Hermal, Kyowa-Kirin, Recordati; Honoraria for lectures/advice: Bristol-MyersSquibb, Novartis, MSD, Almirall-Hermal, Merck-Serono, SUN Pharma, Pierre-Fabre, Sanofi/Regeneron, Immunocore, and Delcath; Meeting support: Pierre-Fabre, SUN Pharma. FW did not have conflicts of interest to disclose. LS has received speakers and/or advisory board honoraria from MSD Sharp&Dohme, Almirall Hermal, Boehringer Ingelheim, Sanofi, UCB, Pfizer, Abbvie, LEO-Pharma. TH has received honoraria as an advisor, speaker and/or investigator from AbbVie, Almirall and Janssen. AK, SD and SS are employees of Almirall Hermal GmbH. DT has received honoraria as an advisor, speaker and/or investigator from AbbVie, Amgen, Almirall, Bristol-Myers-Squibb, Boehringer Ingelheim, Celltrion, Fresenius, Eli Lilly and Company, Galderma, Genzyme, Johnsson& Johnsson, LEO Pharma, New Bridge, Novartis, Pfizer, Regeneron, La Roche-Posay Sanofi, Samsung, Sun Pharma, UCB and Vichy.